Tag: Synthese Chemie
Anwenderkurse zur Peptid-Synthese im Februar 2024
Die CEM GmbH bietet 2024 Ausbildungs- und Anwenderkurse zur Peptid-Synthese in den eigenen Firmenräumen in Kamp-Lintfort am Niederrhein an.
Im Rahmen dieser Kurse werden die Teilnehmer/innen in den theoretischen Grundlagen und insbesondere in der Methodenentwicklung für die Peptid-Synthese geschult. Bei den praktischen Übungen in kleinen Gruppen können die Teilnehmer/innen anhand der individuellen Problemstellungen Methoden programmieren. Das Ziel dieses Anwenderkurses ist das eigenständige Entwickeln von Synthese-Protokollen bzw. Synthese-Methoden.
Der Teilnehmerkreis richtet sich an Laboranten/innen, Ingenieure/innen, Chemiker/innen, etc. Der erste Seminartag beginnt um 13.30 Uhr, es kann also vorher die Anreise erfolgen. Am 2. Seminartag endet die Veranstaltung um 14.00 Uhr, so dass anschließend die Heimfahrt angetreten werden kann.
In Abhängig der Zusammensetzung der Teilnehmer*innen findet der Kurs in deutscher oder englischer Sprache statt.
Der Kostenbeitrag für dieses 2tägige Seminar beträgt 1.500 EUR zzgl. MWSt. und beinhaltet:
- umfangreiche Seminarunterlagen
- zwei Mittagessen
- ein gemeinsames Abendessen
- eine Hotelübernachtung
- den Transfer zwischen dem Hotel und den Schulungsräumen
- und den Transfer am Seminarende zum ICE-Bahnhof Duisburg oder zum Flughafen Düsseldorf.
Folgende Seminarinhalte werden behandelt:
Übersicht und Grundlagen der Peptid-Synthese-Methodik
- Sicherheitsaspekte
- Applikationsbeispiele
- Maintenance & Troubleshooting, selbst Hand anlegen
- Praktikumsstart, Einweisung an den Geräten, Tipps
Grundlagen zur Methodenentwicklung
- Grundlagen der Reaktionsbedingungen für die optimale Synthese
- Grundlagen der Mikrowellen-Peptid-Synthese (Kurs 1)
- Grundlagen zu den Modulen Platten, Säulen, SPOT (Kurs 2)
- Alternative Lösemittel
- Harze, Eigenschaften und optimaler Einsatz
Praktische Arbeit
Praktische Tipps zur erfolgreichen Synthesestrategie
Termine:
Kurs 1: Anwenderkurs für die mikrowellenaktivierte Peptid-Synthese (Discover SPS, Liberty, Liberty Blue, Liberty Prime) am 21. & 22. Februar 2024
Kurs 2: Anwenderkurs für die parallele Peptid-Synthese (MultiPep) am 28. & 29. Februar 2024
Anmeldung:
Anmeldung Anwenderkurs Peptidsynthese
Hinweis
Auf Wunsch bieten wir nach individueller Absprache auch Anwenderkurse vor Ort beim Kunden an. Zur Terminabsprache und für Detailinfos kontaktieren Sie bitte den Kursleiter Herrn Sengutta unter ulf.sengutta(at)cem.com
European Peptide Synthesis Conference 2024
Wir freuen uns, Ihnen mitteilen zu können, dass die European Peptide Synthesis Conference im Jahr 2024 in Prag zurückkehren wird! Egal, ob Sie ein neuer oder erfahrener Peptidchemiker sind, verpassen Sie nicht diese Gelegenheit, sich Experten anzuschließen und sich mit der Peptidsynthese-Community zu vernetzen.
Wir werden bald Details bekannt geben – bleiben Sie dran! #EPSC2024 #PeptideSynthesis #PeptideResearch #Peptidsynthese
Der Einsatz von #Mikrowellenstrahlung zum Energieeintrag für chemische Reaktionen hat sich in den letzten Jahren besonders in der organischen, pharmazeutischen und #Polymerchemie zu einer wichtigen #Synthesetechnik etabliert. Die Hauptvorteile des Einsatzes von #Mikrowellengeräten gegenüber traditionellen Heizquellen sind größere Reaktionsgeschwindigkeiten, höhere Ausbeuten und reinere Produkte. Da moderne #Mikrowellenreaktoren sowohl für #Druckreaktionen als auch für Reaktionen unter #Normaldruck eingesetzt werden können, bieten sie dem #Synthetiker eine Vielzahl von Möglichkeiten zur Durchführung verschiedenster mikrowellenunterstützter Synthesen.
https://analyticalscience.wiley.com/do/10.1002/gitfach.5063/
#chemischeSynthese #Mikrowellensynthese #Nanopartikelsynthese #Polymersynthese #Peptidsynthese #organischeSynthese #anorganischeSynthese
LABO PRODUKT DES JAHRES 2022
Die Laborbranche steckt voller Innovationen, es wird Zeit, diese zu zeigen: Die Leser von LABO wählten im Herbst 2022 die LABO Produkte des Jahres.
In der Kategorie „Life Sciences“ gewann das innovative mikrowellenbeschleunigte Liberty Prime 2.0 Peptidsynthese-System den 1. Platz!
Das Liberty Prime 2.0 ermöglicht die schnelle Synthese von reinen Peptiden und schwierigen Sequenzen in wenigen Minuten satt in vielen Tagen wie bei klassischen Synthesizern. Die neue Liberty Prime Technologie ermöglicht einen 2 min. Kupplungszyklus und extrem schnelle Reagenzienzuführung. Zudem werden bis zu 95 % Lösemittel gegenüber klassischen Peptid-Synthesizern und Mikrowellen-Synthesizern der 1. Generation eingespart.
#GreenChemistry #lifescience #Peptidsynthese #Labo
European Peptide Synthesis Conference 2023
CEM freut sich darauf, die European Peptide Synthesis Conference 2023 im Royal Armouries Museum in Leeds, Großbritannien, vom 28. Februar bis 2. März 2023 zu unterstützen. Begleiten Sie uns drei Tage voller spannender Vorträge über die Synthese von Peptiden und die damit verbundene Forschung. #EPSC2023 bietet eine großartige Gelegenheit für Wissenschaftler und Wissenschaftler in der Peptidbranche, sich an einem einzigartigen Ort und bei einer einzigartigen Veranstaltung zu treffen und auszutauschen.
Erfahren Sie mehr Details und melden Sie sich hier für die Veranstaltung an: https://www.eupeptidesynthesis.com/
#PeptideSynthesis #Peptides #Research #Peptidsynthese
Vor kurzem hat die American Peptide Society eine Reihe von E-Seminaren durchgeführt. Der Vortrag von Jon Collins über optimierte Synthese- und Reinigungsstrategien für Peptide gibt neue Perspektiven in der Peptid-Synthese sowie in der Aufreinigung.
Ein weiterer Höhepunkt der Vortrag zur schnellen Produktion von Neoantigenen, synthetischen Proteinen und das Scale-up der mikrowellengestützten Peptidsynthese auf Hunderte von mmol. Dieses Seminar beinhaltete auch Vorträge von Prof. Dr. Fernando Albericio und einen Vortrag vom Doktoranden Chih-Te „Ted“ Zee.
#Peptidsynthese #Peptidaufreinigung #Prodigy #Liberty
Neues Jahr – Neuer Podcast!
Begleiten Sie Dr. Wendy Hartsock und andere Wissenschaftler in einer neuen Podcast-Serie über Peptide und Proteine. Die Gäste berichten von ihrer Forschung und Sie können von überall (sogar im Labor!) teilnehmen.
Microwaves – „Yesterday“ Cover with Lyrics
So what does Microwave Digestion have to do with Paul McCartney? Most would say not much, but researchers at Abilene Christian University Department of Biochemistry have changed that. Watch these researchers express the artistic side of chemistry as they cover The Beatles song „Yesterday“ with their own twist.
https://www.youtube.com/watch?v=800Yrv1Fwjk
#Mikrowellensynthese #Mikrowelle
During the seminar we will discuss full scale flexibility of automative peptide synthesizers from CEM. We will present how to create peptide libraries for different screening projects: epitope mapping, receptor-ligand interactions, substitutional and lengh analysis. We will also present new peptide purification system, which will simplify and speed up the purification process of hydrophobic and long peptides, proteins. In the end we will learn how to use new green solvents for SPPS.
Mikrowellen-Synthese mit ganz viel Power im Discover 2.0
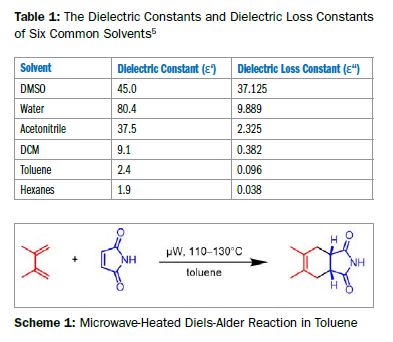
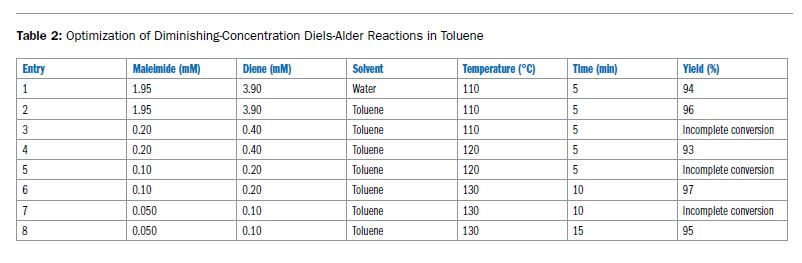
Ein häufiges Missverständnis ist, dass Mikrowellen-Synthesen ein polares Lösungsmittel benötigen. Polare Reagenzien/Edukte/Zwischenstufen interagieren im Discover 2.0 stark genug, um auch in unpolaren Lösungsmitteln effektiv zu erwärmen.
Sehen Sie hier, wie eine mikrowellenbeschleunigte Diels-Alder-Reaktion in Toluol eine hervorragende Ausbeute liefert.
Lesen Sie hier den kompletten Bericht: ApNote-Microwave-Chemistry-with-Non-polar-Reaction-Solutions-ap0186